Category: IGCSE Biology posts

CORMS is dead: Long live DORIC
In Ancient Greece, the Doric order was one of their favoured architectural styles and offered the simplest way of decorating columns.. As you can see from the featured image, Doric columns have an undecorated square capital at their top.
I am sure you won’t get questions on ancient architectural styles in your GCSE Biology, but Doric (now DORIC) is perhaps a simpler way of remembering the key points to include in the experimental design questions in the exam. I have written about CORMS before and there is nothing new in this post, simply a new acronym.
D stands for Dependent Variable. This is what you will measure in your experiment. The mark is often for how you plan to measure the dependent variable, how frequently you will take measurements etc.
O stands for Organisms. What are the key variables relating to the organisms involved? If using a living organism in your experiment (other than humans) often this involves using organisms of the same species, the same age and sometimes the same mass. If you are using humans, you often need to standardise your groups for gender, health, age etc.
R stands for Repeats. If you do more than one replicate of each set of conditions, it allows you to see how reliable your method is and also allows an average result to be calculated. Think about how many repeats you think you would do: it depends on the experiment of course. In a laboratory experiment, three might be sensible, if you are growing seeds to investigate germination, you might grow 200 identical seeds in a tray…..
I stands for Independent Variable. This is the thing you are going to alter in the experiment. So how do you intend to alter it and over what range?
C stands for Control Variables: what are the variables that need keeping the same in every experiment in order to make the investigation a fair test? Think what other factors might affect the dependent variable other than the one you are investigating. And then think how you would keep them constant in an experiment. I would suggest you need to identify at least two or three of the most obvious control variables to ensure you get full marks.
Simple! DORIC is the new CORMS….
Strive for Progress not Perfection


How to revise in the holidays: some PMGBiology tips for 2017….
 Sometimes the hardest thing with revision is getting started….. This post is meant to help you think about how best to make the most difficult first steps towards securing your A* grade. I went through this with my Y11 classes before the end of term so this is just a re-cap. There are very few original thoughts here (story of my life….) but perhaps I can inspire you to get started early in the holidays with your Biology revision….?
Sometimes the hardest thing with revision is getting started….. This post is meant to help you think about how best to make the most difficult first steps towards securing your A* grade. I went through this with my Y11 classes before the end of term so this is just a re-cap. There are very few original thoughts here (story of my life….) but perhaps I can inspire you to get started early in the holidays with your Biology revision….?
Most important – have a plan for the Easter holidays
Work out how many days you have available to revise over the holidays. This will not be the same as the total number of days of holiday as you should have rest days where you do no work at all. Ask your parents what family commitments you have coming up, think about your social life and subtract the days when it will be impossible for you to work. This gives you a number of “working days”.
What do you do on a “working day”?
I suggest that you never try to do more than 4-5 hours of work in any one day. Revision is not measured in hours, it is measured in progress and learning. 2 hours of effective work might be better than 10 hours inefficient time at your desk. The aim here is to maximise the benefit you gain from your revision, not simply clocking up the hours.
Here is how I would organise things if it were me….. Divide the working day into three sessions:
- Morning: 9.30am – 12pm
- Afternoon: 2pm – 4.30pm
- Evening: 7pm – 9.30pm
The plan is this. On a working day you always work in the morning session, every day, no excuses…. Ask your parents to be ruthless in waking you up and don’t allow yourself ever to sleep later than 8.30am or so. There will be plenty of time for lie-ins and getting up at lunchtime in the summer holidays. By midday, you will always have done half the work of the day. How happy will that make you feel? Then choose either the afternoon or evening session (but never both) depending on how you feel and what other plans you have for the day.
How to organise a “revision session”


You need a kitchen timer like the one shown above (not to scale as the textbook is quite large and the timer is quite small…..). I bet if you ask nicely your mum or dad will let you borrow the one in the kitchen. For Biology work, you also need your textbook and revision notes. This is how I suggest you work. Set the timer to 25 minutes, switch off your phone and start work. Work at a topic until the buzzer sounds – no distractions allowed…. If your phone beeps, ignore it. Snapchat can wait! After 25 minutes stop and have 5 minutes off. Make a cup of tea, check your phone and repeat.
I suggest that you revise 5 different subjects in one session. (5 x 25 minutes) You must try to make your revision interesting so you don’t get bored. Bored people do not learn anything……
Different people learn best in different ways so do what works for you. What works for no-one is just reading…. Make notes, revision cards, write out definitions of key facts, use IT if that rocks your boat, whatever you like just do not sit and read your textbook. The key thing is to go over ideas as many times as possible, test yourself on your recall of facts and then try to practise some past paper questions. Most importantly, try to have fun! If you are enjoying it, you will be learning and that is the idea I guess.
Finally, the main benefit of having a plan is this….. When you are not supposed to be working (according to your beautifully crafted plan), you can switch off properly without feeling even a teeny bit guilty. You can combine revising with getting on with enjoying your life. The two things are not mutually exclusive!! And with luck you will avoid feeling like Stewie in the picture below. If only he had a proper revision plan…..


Human Influences on Greenhouse Effect: Grade 9 understanding for IGCSE Biology 4.13 4.14 4.15
I established in a previous post that the Greenhouse Effect is a good thing for life on the planet. So what is the problem? Well the simple idea is that human activities and the massive growth in human populations seen over the past two hundred years have changed the composition of the atmosphere. The concentration of greenhouse gases has risen and this enhanced greenhouse effect is causing climate change.
The principal gases in the atmosphere responsible for the greenhouse effect are carbon dioxide, methane and water vapour. Have a look at these two tables taken from the following website from the Center for Climate and Energy solutions:
http://www.c2es.org/facts-figures/main-ghgs
The first image shows the main gases in the atmosphere that contribute to the Greenhouse Effect.

Anthropogenic means “caused by mankind” and so you can see what humans are doing to generate an enhanced greenhouse effect. The GWP figure stands for Global Warming Potential and gives a relative value for how each gas might contribute to climate change. One molecule of CFC-12 is as powerful as a greenhouse agent as 10,900 molecules of carbon dioxide.

This second table shown above demonstrates how the composition of the atmosphere has changed from pre-indutrial to modern times. I am going to focus on the two greenhouse gases at the top of the list: carbon dioxide and methane.
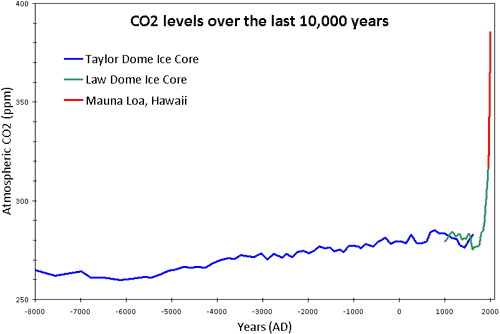
Carbon Dioxide concentrations in the atmosphere
Scientists at the Maunua Loa Observatory in Hawaii have been measuring atmospheric carbon dioxide concentrations since the 1950s. Here is a graph of their results.
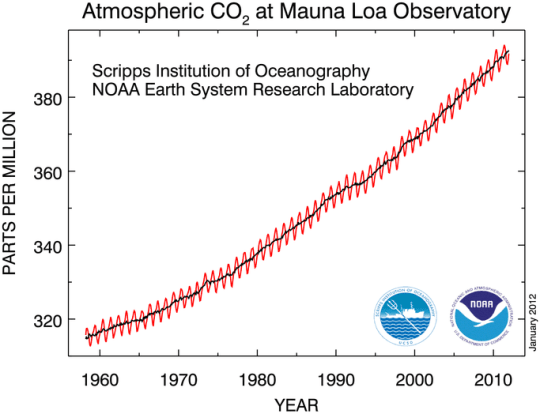
What do you notice about this graph?
- There is a gradual upward trend such that the average concentration has risen steadily over the 50 year period.
- Within each year, there is an annual peak and an annual trough in the carbon dioxide concentration. The “peak” corresponds to northern hemisphere winters when there is less photosynthesis by plants and more fossil fuels are burned. The “trough” is northern hemispere summer when photosynthesis rates are high and so carbon dioxide is removed from the atmosphere
If you want data going back further into the past, you need to look at ice core data. Tiny volumes of the atmospheric gases are trapped within ice as it forms in Antartica and by drilling out a core and analysing the gases it contains, one can determine the concentration of the atmosphere when the ice was formed. The deeper parts of the core formed longer ago so a journey through an ice core is like travelling back in time…..
What human activities might be responsible for these changes in carbon dioxide?
- Deforestation (see my post on this topic)
- Combustion of Fossil Fuels (coal, oil, gas)
- Cement production
Methane concentrations in the atmosphere

Methane is also a potent greenhouse gas. It is produced as waste product of the bacterial reactions that happen inside the rumen and intestines of cattle. (The rumen is the large first chamber of their stomach in which bacteria digest cellulose in the cow’s food) I was once told that each cow produces 65 litres of methane a day but I have never measured it myself……. Seeing as the world population of cattle is estimated at 1.4 billion, that is a lot of methane each day being released into the atmosphere.
Methane is also produced by bacteria that break down our domestic waste in land fill sites and by anaerobic bacteria that live in paddy fields in which rice is grown. More humans means more cattle, more rice and more landfill and all of these are responsible for the rise in methane concentrations seen in the atmosphere in recent times.

Greenhouse Effect: Grade 9 understanding for IGCSE Biology 4.13 4.14 4.15
The Greenhouse Effect is the name given to the way in which the earth’s atmosphere acts to warm up the planet. The earth and the moon gain almost exactly the same incident radiation from the sun and yet average temperature on the earth is stable at around 14 degrees Celsius. On the moon the temperature fluctuates wildly from 1oo degrees Celsius during the day to minus 153 degrees Celsius at night. Life would be impossible in such extreme and variable conditions and so the greenhouse effect is definitely a “good thing” for life on our planet.
How does the greenhouse effect work?
Well the main idea here is that there are certain gases in the atmosphere that can trap the infrared radiation that the earth emits and prevent it escaping the atmosphere. These greenhouse gases are warmed as they absorb the infrared and so the atmosphere heats up.
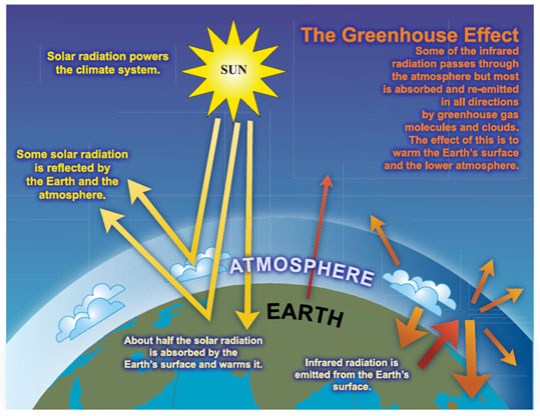
Remember that because the sun is so hot, it emits radiation at a much higher frequency. This is mostly in the “visible light” part of the spectrum together with some ultraviolet. The gases in the atmosphere cannot trap visible light (air is transparent as you have probably noticed) and so most of the solar radiation passes through the atmosphere and hits the earth.

Which gases can act as greenhouse gases?
The two most prevalent gases in the atmosphere are nitrogen (N2) and oxygen (O2) and neither is able to trap infrared so cannot act as a greenhouse gas. The principle greenhouse gases in the atmosphere are

- carbon dioxide
- water vapour
- methane
- ozone
- nitrous oxide
So what’s the problem?
Well of course the problem is that human activities over the past century or so have altered the composition of the atmosphere so that the concentration of greenhouse gases has risen. This has meant more heat is trapped and climate systems are altered in consequence. This enhanced-greenhouse effect is the problem and I will look at this in the next post…..


Evidence for Climate Change: Grade 9 understanding for IGCSE Biology (part 2) 4.13 4.14 4.15
Before you can look at the science of climate change and how human activities are causing it, you first need to accept that we are currently undergoing a period in which our climate is changing at an unprecedented and rapid rate.
What would you expect to see in a warming world?
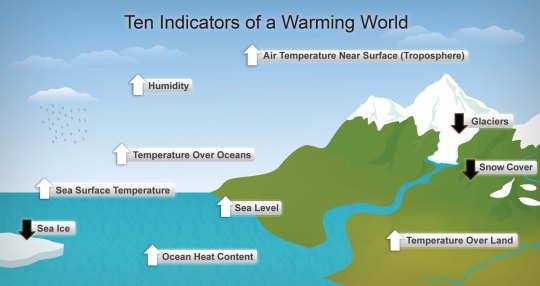
Well the first and most obvious point is that you would expect to see measurable changes in land and sea temperatures.
The top ten hottest years on record have all happened since 1998.
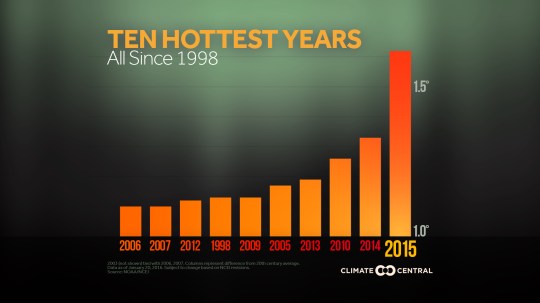
There is excellent evidence from all over the planet of glaciers shrinking; polar ice caps are shrinking and it may be that the North Pole is free of summer sea ice for the first time in 100,000 years some time in the coming decades. (This is controversial as it is a prediction based on computer models of future climate: different models make different predictions and with a system as chaotic and interwoven as global climate, it can be very difficult to predict)
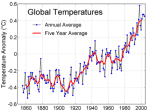
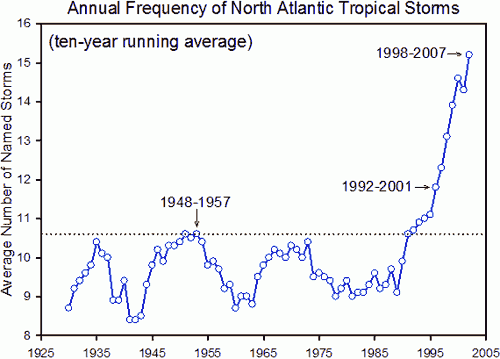
As the oceans have warmed, this has impacted on extreme weather systems. This graph shows the incidence of North Atlantic Tropical Storms over the past century or so. It has continued to rise since 2007…..
When you look at all this evidence, it is hard to believe that our climate has been stable over the past century. In fact very few people try to dispute the fact of climate change. The dispute is whether human activities are causing this climate change and indeed whether it matters….. I hope in later posts I can convince you the answers to these two questions are yes and yes.

Air pollution and climate change: Grade 9 understanding for IGCSE Biology (part 1) 4.13 4.14 4.15
Climate change remains one of the more controversial topics in the IGCSE Biology specification. Just in the past few weeks, the USA (one of the finest nations on the planet) has elected as their President someone who has stated on record that he believes in some giant conspiracy theory about climate science centred around the Chinese….

The overwhelming majority of climate scientists do not support this interpretation of the facts. They are able to provide evidence of rapid climate change over the past few decades and link this to human-induced changes in the composition of the atmosphere due to pollution. When these facts are linked by a sensible scientific theory that proposes how and why certain gases might lead to an increased warming effect in the atmosphere (the so-called greenhouse effect) the evidence in support of human-induced climate change becomes compelling. Keeping US manufacturing competitive is important of course, but not at the expense of the enormous environmental and financial costs of allowing our pollution of the atmosphere to continue unchecked. I am not sure I will be able to convince President-Elect Trump (he probably doesn’t read my blog in any case) but perhaps I can show you the kind of understanding needed to generate A* answers in GCSE questions on this topic…?

I am going to organise this work into several sections and will post on each topic in the coming week….
- What is the evidence for climate change?
- What is the greenhouse effect?
- How are human activities altering the make up of the atmosphere?
- What are the predicted consequences of climate change in the coming years?
Bacteria and Yoghurt: Grade 9 understanding for IGCSE Biology 5.7
Biology is a great subject to teach: you get to introduce young minds to the wonders of the natural world, to show them how evolution has been able to take an ancient planet with a few self-replicating molecules in some deep sea vent and end up today with perhaps 100 million different species all occupying unique niches in an ever changing ecosystem. You can take students onto a journey into the cell so that they understand and appreciate the complexities of DNA as a coding molecule and how proteins have evolved to carry out the myriad processes of cellular biology.
And you also get to teach the role of bacteria in yoghurt making. This will be a short post.
This is yoghurt. It is made from milk.

Milk contains a sugar, lactose. Yoghurt is made when a culture of Lactobacillus bacteria is added to the milk. The two most important bacterial species involved in yoghurt making are called Lactobacillus bulgaricus and Streptococcus thermophilus (although only the former is mentioned in the specification). Perhaps they didn’t want the excitement levels to get too high….?
Lactobacillus use the lactose in the milk as a respiratory substrate producing lactic acid as a waste product. This lactic acid gives the yoghurt its bitter taste as well as lowering the pH so that the yoghurt takes on its gel like appearance.
Respiration in Lactobacillus:
Lactose ——> Lactic Acid
That’s the limit of my knowledge and interest in this topic: hope it helps…….
Yeast and Brewing: Grade 9 Understanding for IGCSE Biology 5.5
Humans first discovered how to make beer around 7000 years ago and brewing has been an integral part of human civilisations ever since. Yeasts are a family of single celled fungi that can use the sugars in fruits and seeds as a source of energy for respiration. Yeast can respire both aerobically and anaerobically and you should know the equations for these two processes.
Aerobic respiration
Glucose + Oxygen ——-> Carbon Dioxide + Water
Anaerobic respiration (aka Fermentation)
Glucose ——> Ethanol + Carbon Dioxide

So when yeasts respire anaerobically they produce ethanol and carbon dioxide as the waste products. Ethanol is also known as alcohol and humans learned a long time ago that alcohol is a drug that changes the way you think or feel, often in a pleasurable way in moderate doses. Making drinks that were alcoholic also helped to kill potentially harmful bacteria and other pathogens in pre-industrial times when drinking water was not readily available.
If you add yeast to a source of sugar in anaerobic conditions, the yeast cells will ferment the sugars into alcohol and carbon dioxide. In order to make beer, the source of sugar comes from germinating barley seeds. Hops (dried flowers of a hedgerow plant) are added later to give the bitter flavour beer drinkers seem to like…..
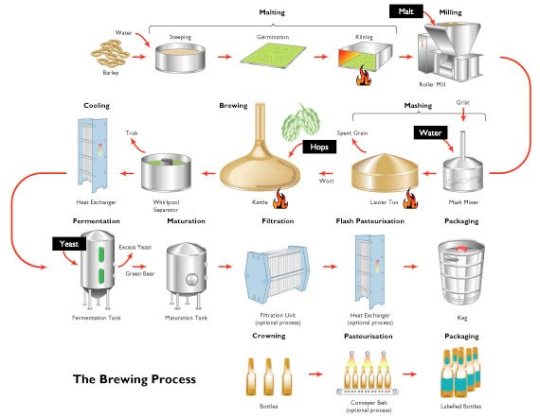
The flow diagram above shows the stages in making beer. I can’t imagine you would be expected to know the details. Any question on this topic would presumably focus on the anaerobic respiration of the yeast rather than the details of the brewing process.
In case anyone is interested, I am not a huge fan of beer although I can occasionally be forced by peer pressure into consuming one or two. My preferred fermentation reactions happen not in the copper tuns of English breweries but in the beautiful Northern Rhone valley in France, where skilled wine-makers can take Syrah grapes grown under the influence of the cooling mistral wind and turn them into beautiful Cote Rotie or Hermitage. Now there’s a happy thought for a cold November evening……..


Fertilisers: Grade 9 understanding for IGCSE Biology 5.3
Fertilisers is the term used for “chemicals or natural substances added to soil to promote the growth of plants”.
Key point: in spite of what it says on this packet of fertiliser, fertilisers are not food for plants. (Just adding this photo to the post makes me feel slightly sick inside: how could MiracleGro be so happy to confuse generations of people who visit garden centres…..?)

Plants are autotrophic: they make their own food molecules in the amazing process of photosynthesis. Plants use carbon dioxide from the air plus water from their roots to produce a whole range of organic molecules powered by the energy from sunlight.
But remember that in order to make amino acids, proteins and DNA plants will also need a source of nitrogen atoms. Carbon dioxide and water do not contain any nitrogen atoms and yet nitrogen is needed for building amino acids, proteins and DNA.
Where do plants get this nitrogen from?
Well the key idea is that they do not take it from the air. Nitrogen gas in the air is very un-reactive and cannot be fixed in the plant. But the soil contains nitrate ions and plants can absorb these by active transport in their root hair cells. Nitrate ions are transported up the plant in the xylem and can be used to make amino acids etc. in the leaf cells.
Nitrates are not the only mineral ions taken up by plants in their roots. Plants absorb phosphate (for making DNA), magnesium (for making chlorophyll), potassium (for a wider variety of cellular processes) amongst many others.
So fertilisers are a way of replenishing the concentration of these essential minerals in the soil. More fertiliser, more minerals, faster plant growth as more proteins/DNA etc. can be made in the leaves…. Simples!

The commonest type of inorganic (chemical) fertiliser are called NPK fertilisers. (Nitrogen, Phosphate, Potassium). These can be bought in handy 50kg sacks (see picture above), stored and then spread easily over fields.
Farmers can also use manure which is an organic fertiliser. Here are some advantages/disadvantages of organic fertilisers in case you are interested…. It is smelly, bulky and difficult to store.