Tagged: fermentation
Yeast and Brewing: Grade 9 Understanding for IGCSE Biology 5.5
Humans first discovered how to make beer around 7000 years ago and brewing has been an integral part of human civilisations ever since. Yeasts are a family of single celled fungi that can use the sugars in fruits and seeds as a source of energy for respiration. Yeast can respire both aerobically and anaerobically and you should know the equations for these two processes.
Aerobic respiration
Glucose + Oxygen ——-> Carbon Dioxide + Water
Anaerobic respiration (aka Fermentation)
Glucose ——> Ethanol + Carbon Dioxide

So when yeasts respire anaerobically they produce ethanol and carbon dioxide as the waste products. Ethanol is also known as alcohol and humans learned a long time ago that alcohol is a drug that changes the way you think or feel, often in a pleasurable way in moderate doses. Making drinks that were alcoholic also helped to kill potentially harmful bacteria and other pathogens in pre-industrial times when drinking water was not readily available.
If you add yeast to a source of sugar in anaerobic conditions, the yeast cells will ferment the sugars into alcohol and carbon dioxide. In order to make beer, the source of sugar comes from germinating barley seeds. Hops (dried flowers of a hedgerow plant) are added later to give the bitter flavour beer drinkers seem to like…..
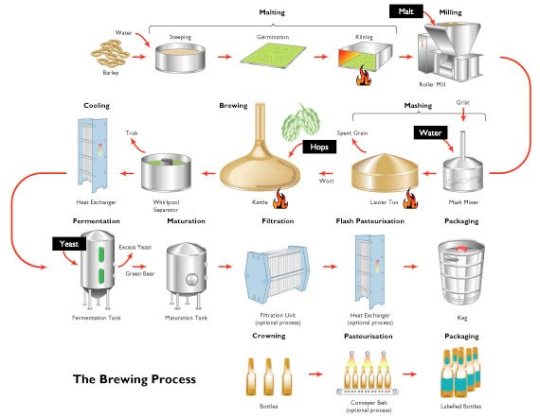
The flow diagram above shows the stages in making beer. I can’t imagine you would be expected to know the details. Any question on this topic would presumably focus on the anaerobic respiration of the yeast rather than the details of the brewing process.
In case anyone is interested, I am not a huge fan of beer although I can occasionally be forced by peer pressure into consuming one or two. My preferred fermentation reactions happen not in the copper tuns of English breweries but in the beautiful Northern Rhone valley in France, where skilled wine-makers can take Syrah grapes grown under the influence of the cooling mistral wind and turn them into beautiful Cote Rotie or Hermitage. Now there’s a happy thought for a cold November evening……..


Demonstrating Carbon Dioxide production in Respiration of Yeast 2.39
Yeast is a single celled fungus that can respire aerobically when oxygen is available and anaerobically in the absence of oxygen.
Aerobic respiration in yeast:
Glucose + Oxygen ======> Carbon dioxide + Water
Anaerobic respiration in yeast:
Glucose ======> Ethanol and Carbon dioxide
Both forms of respiration produce carbon dioxide as a waste product so how could this be demonstrated experimentally?
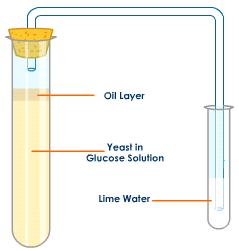
This is the simplest set up that could demonstrate this. Lime water will go cloudy in the presence of carbon dioxide. Glucose solution is needed to provide the reactant sugar for the yeast to respire. The oil layer on the top is to prevent the diffusion of oxygen from the air into the Yeast in Glucose solution, this ensuring anaerobic respiration will occur.
What experimental factors could be altered in this set up?
Well assuming you keep the volume and concentration of lime water constant, the time taken for the limewater to go cloudy could be measured under differing conditions: the faster the time, the faster the rate of respiration. The experimenter could investigate the effect of changing the temperature, the pH or the concentration of glucose solution used. Make sure you understand how and why changing each of these factors might affect rates of respiration in yeast.