Demonstrating Carbon Dioxide production in Respiration of Yeast 2.39
Yeast is a single celled fungus that can respire aerobically when oxygen is available and anaerobically in the absence of oxygen.
Aerobic respiration in yeast:
Glucose + Oxygen ======> Carbon dioxide + Water
Anaerobic respiration in yeast:
Glucose ======> Ethanol and Carbon dioxide
Both forms of respiration produce carbon dioxide as a waste product so how could this be demonstrated experimentally?
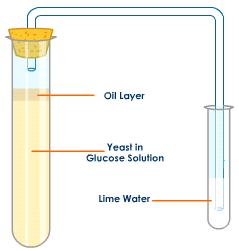
This is the simplest set up that could demonstrate this. Lime water will go cloudy in the presence of carbon dioxide. Glucose solution is needed to provide the reactant sugar for the yeast to respire. The oil layer on the top is to prevent the diffusion of oxygen from the air into the Yeast in Glucose solution, this ensuring anaerobic respiration will occur.
What experimental factors could be altered in this set up?
Well assuming you keep the volume and concentration of lime water constant, the time taken for the limewater to go cloudy could be measured under differing conditions: the faster the time, the faster the rate of respiration. The experimenter could investigate the effect of changing the temperature, the pH or the concentration of glucose solution used. Make sure you understand how and why changing each of these factors might affect rates of respiration in yeast.
what if in an examination one is asked why the glucose solution was boiled before adding yeast
boiling removes the gases present in the water before yeast is added.
(this is probably too late and you must have figured the answer out already)
The CO2 will form bubbles that will rise to the top of the oil. This creates a one way gravitational barrier that lets any gas bubbles go up, but not down. The gas composition is not relevant to the operation of the oil to act as a one way barrier. I hope that helps to understand the operation of the oil.
Science is Everything, Know your Universe!
Hi, I don’t understand how the carbon dioxide gets into the tube – surely the oil on top of the yeast solution not only keeps oxygen out, but also keeps carbon dioxide in? So why doesn’t the little tube have to go down into the solution?
Good question. I guess the concentration gradient for carbon dioxide is so steep that it can diffuse across the oil layer. Or perhaps the oil layer is more permeable to carbon dioxide than to oxygen (but I am not a good enough chemist to see why this might be) You can’t put the little tube into the solution because you need carbon dioxide gas, not dissolved carbon dioxide, to increase the air pressure inside the boiling tube.
why is carbon dioxide still able to pass through the top layer of the oil
This is a good question. I think this question has been raised before so have a read of the comments under the post. I think the short answer would be that what you are told is something of a simplification. You really need to boil the glucose solution to remove all dissolved gases. But that still doesn’t really make sense for the reason you give in your question….. If you are interested, I would suggest talking to a chemistry teacher at school and see if they can give a sensible reason for this. If you find anything out please do add it as a comment to this post!