Tagged: pepsin

Digestion of Proteins: Grade 9 Understanding for IGCSE Biology 2.29
Proteins are large insoluble molecules made up of many hundreds of amino acids joined together in a long chain. So in order to obtain these molecules from our diet, the large protein must be digested (broken down) into the smaller amino acid subunits. Amino acids can be absorbed into the blood stream in the ileum, part of the small intestine.
The family of enzymes that can catalyst the digestion of proteins are called proteases.
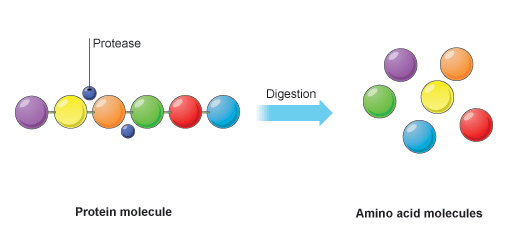
Protein digestion happens in a two-stage process. In the first stage the large protein molecules are broken down into smaller proteins (often called polypeptides) by a protease enzyme. Pepsin is one such protease and acts in the stomach.
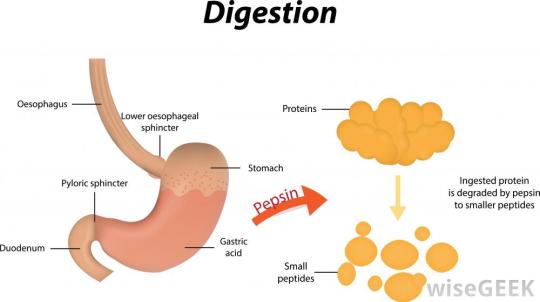
Remember that the food in the stomach is mixed with hydrochloric acid. This results in a very acidic liquid in the stomach (chyme). Pepsin works in the stomach and so rather unusually for a digestive enzyme, it has an optimum pH of pH 1.5 – pH2.
The second protease enzyme that you should know about is trypsin. Trypsin is made in the pancreas and so enters the duodenum soon after the stomach contents pass the pyloric sphincter (see diagram above). The acidic chyme that enters the duodenum is rapidly neutralised by hydrogencarbonate ions (an alkali) secreted in the bile and in pancreatic juice. Trypsin has an optimum pH of around pH 7.5.
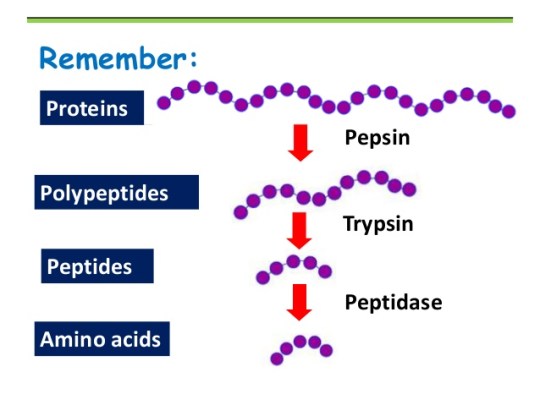
As shown in the diagram above, there is a final stage to protein digestion. The actions of pepsin in the stomach and trypsin the duodenum result in small protein fragments called peptides. Many peptides are still too large to be absorbed into the blood in the ileum and so need digesting further into their constituent amino acids. Peptidase enzymes are embedded in the epithelial cell membranes in the small intestine and this final reaction completes the digestion of proteins.
Amino acids are absorbed by active transport into the blood capillaries in the villi in the small intestine.