Tagged: lipids
Digestion of Lipids: Grade 9 Understanding for IGCSE Biology 2.29 2.30 2.31
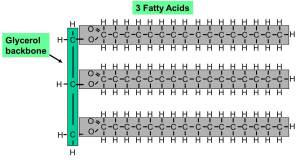
A balanced diet will contain many different fats and oils. The commonest type of molecule in these lipids is called a triglyceride. They are made from a small molecule of glycerol attached to three fatty acids.
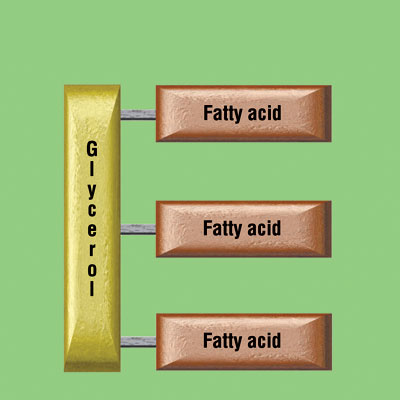
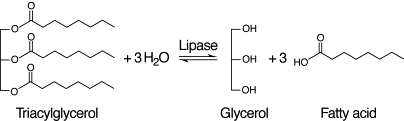
These triglycerides are too large to be absorbed in the small intestine (ileum) and so need to be broken down into their constituent parts. In the digestive system there are enzymes called lipases that can catalyse the digestion of lipids into fatty acids and glycerol.
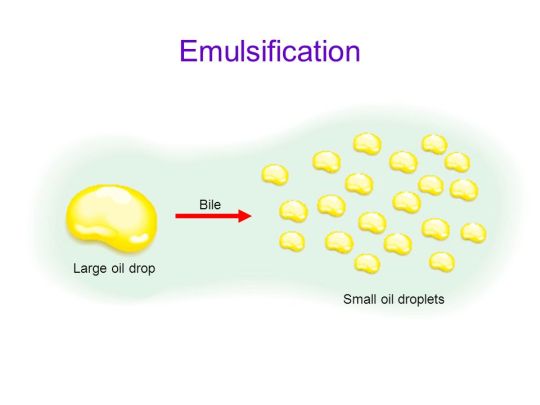
Most digestion of lipids happens in the duodenum. The pancreas produces a lipase enzyme that mixes with the food in the duodenum. Although bile does not contain any digestive enzymes, it does have bile salts that play an important role in the digestion of lipids. Bile salts cause large fat droplets in the duodenum to change into many smaller lipid droplets – a process called emulsification. (Read my post on bile if you want more details here….)
Fatty acids and glycerol molecules are small enough to cross the epithelium in the villus in the ileum. This absorption of fatty acids and glycerol is slightly different to the other products of digestion as they do not pass immediately into the blood. They are assembled immediately into structures called chylomicrons and these move into the single, blind-ended tube in the villus called a lacteal. The lacteals merge together into lymph vessels that eventually empty into the blood in the neck. (Please read my post on absorption in the small intestine to read more about this)

Biological Molecules: Grade 9 Understanding for IGCSE Biology 2.7 2.8
You will have studied the Biological Molecules section in some detail I would imagine, perhaps in more detail than is absolutely required for the specification. This post is meant to help you focus your understanding onto those points that are most likely to be tested in iGCSE questions. Here goes…
You do need to understand some chemistry for this topic to make sense. In particular you need to understand what is meant by the following terms:
- atom
- molecule
- element
- compound
My personal definitions would be as follows:
Atom: the smallest particle that retains the chemical properties of the element – a structure made up of protons, neutrons and electrons
Molecule: a particle made of two or more atoms chemically bonded together – may contain just one type of atom or several
Element: a substance in which all the atoms are the same
Compound: a substance containing more than one type of element
Back to safer ground…..
Living organisms are made from a fairly small group of molecules. The commonest molecule in every organism is water and in humans water makes up about 70% of the mass. But if you were to remove all water, leaving behind just the dry mass, the most common molecules could be grouped into proteins, lipids, carbohydrates and nucleic acids (e.g DNA)
Carbohydrates contain just three elements – carbon, hydrogen and oxygen
Lipids (fats and oils) contain three elements – carbon, hydrogen and oxygen
Proteins contain four or five elements – carbon, hydrogen, oxygen, nitrogen and sometimes sulphur
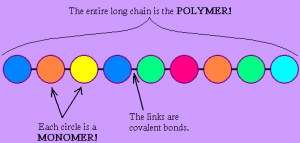
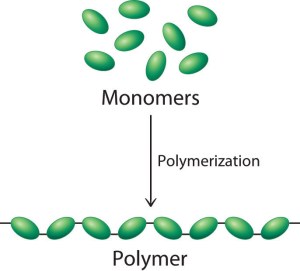
Big idea: many of the molecules that living things are made from are examples of polymers. A polymer is a large molecule made up of a long chain of repeating subunits (called monomers)
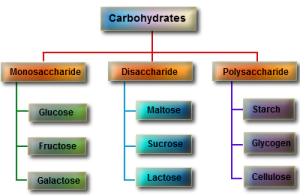
Carbohydrates are grouped into three main types:
Simple sugars like glucose or fructose – these are called monosaccharides.
Some sugars like sucrose are made of two simple sugars joined together – these are called disaccharides
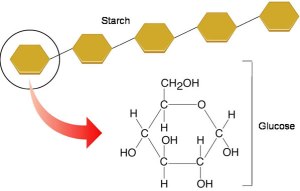
Some carbohydrates are macromolecules (polymers) made of many hundreds of sugar residues joined together – these are called polysaccharides.
You can see from the diagram above that there are three important polysaccharides in living organisms. All three are polymers of the sugar glucose but the arrangement of the glucose residues is different. Cellulose is the main constituent of plant cell walls. Starch is a storage polysaccharide found in plants and Glycogen is a similar storage molecule found in liver and muscle tissue in animals.
Glucose is detected using a Benedict’s Test. Heat the solution with Benedict’s,reagent to 90 degrees for 5 minutes. A positive test for glucose is a brick red colour.
Starch is tested for using iodine solution (in potassium iodide) Iodine solution turns blue-black in the presence of starch.
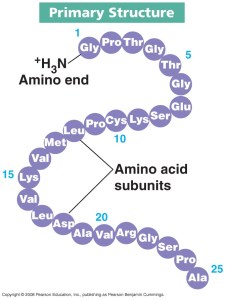
Proteins are also polymers but this time the individual monomer is not a sugar but a molecule called an amino acid.
This protein is then folded up into a complex 3D shape using a whole load of weak bonds that can easily be broken at high temperatures. This is why enzymes, made of protein, denature at high temperatures.
There are 20 different amino acids that could be incorporated into a protein so there are an almost limitless variety of different proteins that can be made.
Lipids are a group of water-repelling molecules that again contain C,H and O atoms. They used to be separated into fats and oils depending in whether they are a solid (fat) or liquid (oil) at room temperature. Many lipids are a type of molecule called a triglyceride and this is made of a single molecule of glycerol attached to three fatty acid tails.