Tagged: fermenter

Recombinant DNA: Grade 9 Understanding for IGCSE Biology 5.12 5.13 5.14
In the last post on this topic, I explained about the two types of enzymes needed for the genetic modification of organisms:
Restriction enzymes that can cut up DNA molecules at specific target sequences, often resulting in fragments with sticky ends
DNA ligase that joins together fragments to form a single DNA molecule
The EdExcel iGCSE syllabus uses the example of the genetic modification of bacteria to produce human insulin. Human insulin is a hormone that helps regulate the concentration of glucose in the blood. It is made in the pancreas when the blood glucose concentration gets too high and causes liver cells to take up glucose from the blood and convert it to the storage molecule glycogen. Patients with type I diabetes cannot make their own insulin and so need to inject it several times a day after meals to ensure they maintain a constant healthy concentration of glucose in their blood.
Bacteria can be genetically modified so that they produce human insulin. These transgenic bacteria can be cultured in a fermenter and the insulin produced can be extracted, purified and sold.
How do you get hold of the human insulin gene?
Well the honest answer is that there are a variety of ways of achieving this. It can now be synthesised artificially as we know the exact base sequence of the gene but it can also be cut out of a human DNA library using a restriction enzyme. There are other ways too but for the sake of brevity (and sanity) I am not going to go into them here. [If you are really interested in this, find out how reverse transcription of messenger RNA from cells in the pancreas can allow you to build the insulin gene.]
How do you get the human insulin gene into a bacterium?
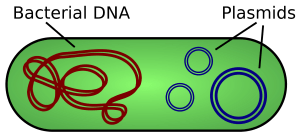
Remember that bacterial cells are fundamentally different to animal and plant cells. One difference is that bacterial cells have no nucleus and their DNA is in the form of a ring that floats in the cytoplasm. Many bacteria also have plasmids which are small additional rings of DNA and these provide a way for getting a new gene into a bacterium.
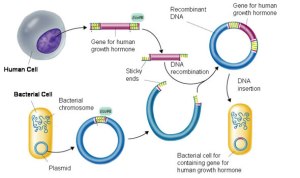
Bacteria exchange plasmids in a process called conjugation and so it is fairly easy to get the plasmid out of the bacterium. If the plasmid is cut open using the same restriction enzyme as was used to cut out the human insulin gene, the sticky ends will match up and so DNA ligase will join the two pieces of DNA together to make a recombinant plasmid. The diagram below shows the process for human growth hormone but it would be exactly the same for the example we are looking at.
If the recombinant plasmids are inserted into bacteria, the bacteria will read the human insulin gene and so produce the protein insulin.
How do you grow the transgenic bacteria on an industrial scale?
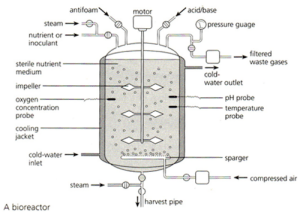
The bacteria that have taken up the recombinant plasmid are grown in a fermenter. This is a large stainless steel vat (easy to clean and sterilise) that often has several design features conserved between different varieties:
The fermenter usually has a cooling jacket to carry away excess heat. The jacket often has a cold water input pipe and the warmer water is carried away. There has to be some mechanism for mixing the contents of the fermenter so the diagram above shows paddles attached to a motor. Fermenters also need a sterile input system for getting air, water and nutrients into the fermenter but without introducing foreign bacteria and fungi. Air is needed as the bacteria are aerobic and need oxygen for respiration.
If the bacteria in the fermenter contain the human insulin gene, then they will be able to produce human insulin. This can be extracted, purified and sold to the NHS for treating type I diabetics.