Tagged: enzyme

Enzymes: Grade 9 Understanding for IGCSE Biology 2.10 2.11 2.13
Enzymes are biological catalysts. This means they are able to increase the rate of a chemical reaction but are not used up in the reaction. Without enzymes the reactions of metabolism would all happen too slowly for life to exist – enzymes can speed up the rate of reaction by many millions of times….
Enzymes work as catalysts by lowering the activation energy needed for the reaction to occur. Activation energy is the term for the extra energy needed to be given to the reactants to break bonds within them to allow the product molecules to be formed. Enzymes provide an alternate reaction pathway that has a lower activation energy. This means that under any conditions a higher proportion of the reactant molecules will have sufficient energy to overcome the activation energy barrier and so more reactants will be turned into products.
How do enzymes lower the activation energy?
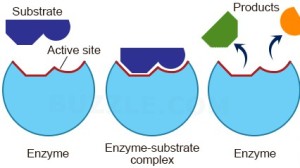
Enzymes are all large globular molecules, almost always made of protein. They have a specific three-dimensional shape that includes a region called the active site which has a shape that allows the reactant molecules (called substrates) to bind. When the enzyme binds to the substrate, it forms an enzyme-substrate complex.
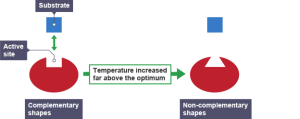
This theory of how enzymes might work is called the Lock and Key theory. The active site acts like a lock as it has a shape that is complementary to the shape of the substrate (the key). Lock and Key theory explains an important property of enzymes which is that they are specific. Each enzyme can only catalyse one reaction since only a substrate molecule with a specific shape can bind to the active site.
When the substrate is bound to the active site forming an enzyme-substrate complex, the enzyme introduces a strain on some of the bonds in the substrate, making a reaction more likely. The active site might provide a microenvironment that is exactly the right condition for the reaction, thus lowering the activation energy.
Key idea: enzymes catalyse almost all the chemical reactions that happen in organisms. It is easy to imagine that enzymes only catalyse reactions like the one in the picture above in which a molecule is being broken down into smaller molecules. But enzymes catalyse oxidation reactions, condensation reactions in which big molecules are built up from smaller ones, phosphorylation reactions (sticking phosphate groups onto molecules) and so on and so on. So don’t describe enzymes as being involved in breaking things down: some do of course but the vast majority work inside cells to catalyse a whole variety of reactions in metabolism.
Rates of enzyme-catalysed reactions can be affected by Temperature
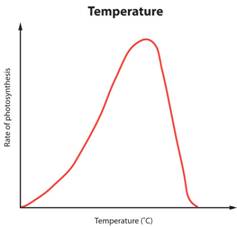
The temperature of the reaction has a significant effect on the rate of reaction. Look at the following graphs:
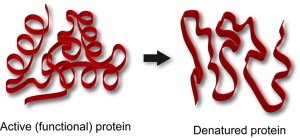
The pattern of this graph is characteristic of an enzyme catalysed reaction. At low temperatures the rate of reaction is low. This is because few enzyme-substrate complexes are formed per second as the enzyme and substrate molecules are moving around so slowly that they rarely collide. At temperatures above the optimum, the enzymes and substrate molecules will be moving very fast and so will be colliding all the time. So why is the rate so low? Well that is because high temperatures cause enzymes to denature. Remember enzymes are made of protein and proteins can have their 3D shape changed by high temperatures. If an enzymes’ 3D shape changes, the active site will change shape and if this happens the substrate cannot bind.
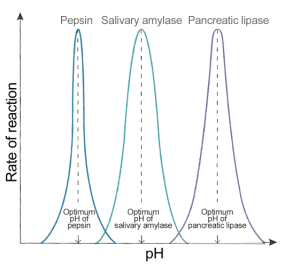
Rates of enzyme-catalysed reaction are also affected by pH
Enzymes tend to work in a very narrow band of pH values. pH is a measure of the acidity/alkalinity of a solution and most enzymes require an optimum pH to function well. The rate of reaction drops very rapidly on either side of the optimum pH simply because extremes of pH will denature enzymes. The acid or alkaline environment can break the bonds that hold the enzyme in its specific 3D shape. An enzyme with a changed shape cannot function as a catalyst if the substrate cannot bind to the active site and so the rate falls away rapidly either side of the optimum.