Tagged: 2.13

Diffusion, Active Transport and Osmosis: Grade 9 Understanding for IGCSE Biology 2.15 2.16
This post is going to describe some of the ways molecules can cross the cell membrane. (For Eton students revising for Trials, diffusion and active transport are found in the F block syllabus, osmosis comes in E Block)
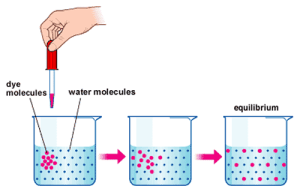
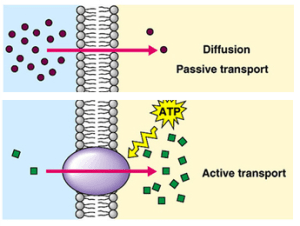
Diffusion is the simplest to understand. Diffusion does not even need a cell membrane to occur. In the example below the dye molecules will move randomly in the solution. As the dye starts in one place, these random movements will mean that slowly spread out until an equilibrium is reached. This movement of the dye from the region of high concentration to the low concentration is called diffusion.
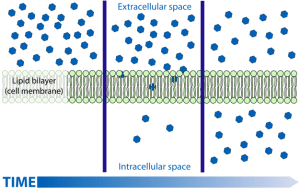
When considering diffusion into a cell, if the cell membrane is permeable to a particular molecule then the random movements of the molecule will mean that there will be a net (overall) movement from the higher concentration to the lower concentration down the concentration gradient.
Key Points about diffusion:
- Always happens down a concentration gradient (from a high concentration to a lower one)
- Never requires any energy from the cell – it is a passive process
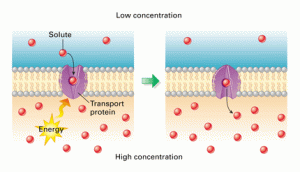
Active Transport is a process that will move molecules into a cell against the concentration gradient – i.e. from a low concentration to a high concentration. This “pumping” of the molecules against the gradient requires energy from the cell and of course this energy comes from respiration.
You can see from the diagram above that active transport is working against the concentration gradient, is using energy from inside the cell (actually a molecule made in mitochondria in respiration called ATP) and that a specific transport protein is involved in the cell membrane. This protein will have a binding-site that is specific for a particular molecule and the solute molecule to be transported will collide with the transport protein due to random movement. Energy from the cell can cause the transport protein to change shape such that the solute is released on the other side of the membrane.
Can you think of another area of the iGCSE syllabus which features collisions between a specific binding-site on a protein and a certain other molecule? Linking ideas is a key characteristic of the A* Biologist!
Osmosis is the hardest of these processes to understand properly, especially as an iGCSE student when you are often told an over-simplified account that does not make sense…. Let’s try to simplify it in a way that does make sense.
Firstly it is only water molecules that can move by osmosis into and out of cells – never anything else. Indeed osmosis is the only way water can cross a membrane – it never moves by diffusion or active transport.
Osmosis is a passive process – it never needs any energy from the cell’s respiration and the only energy involved is the kinetic energy of the water molecules.
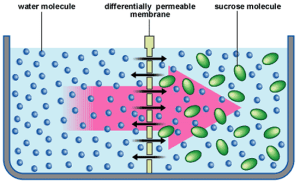
Osmosis can only occur through a partially permeable membrane. All cell membranes are partially permeable and this means they let small molecule like water through but prevent the diffusion of the larger solute molecules.
The water molecules on both sides of the membrane in the diagram above will be moving around randomly. They will occasionally hit one of the pores in the membrane and so pass across the membrane. This movement will be happening from left to right and from right to left.
But….
The presence of the sucrose (solute) in the solution on the right means that some of the water molecules on that side of the membrane are less able to move. This is because they are temporarily attracted to the solute molecules by weak hydrogen bonds. So their kinetic energy is reduced and this makes them less likely to randomly collide with the pores in the membrane. The presence of the solute on the right means that water molecules on the left on average are more likely to collide with the membrane than the water molecules on the right and this leads to an overall movement from left to right. This net movement of water molecules from the dilute solution to the more concentrated solution through the partially permeable membrane is called osmosis.
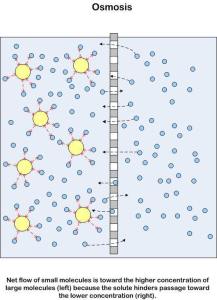
This diagram has the two solutions reversed so in which direction will osmosis happen here? Thats right from right to left. You can see the hydrogen bonds attracting water molecules to the solute – these are the ones that lower their kinetic energy overall.
You might even have been taught about osmosis with reference to the water potential of a solution. The water potential of a solution is just a measure of how much kinetic energy the water molecules in a solution possess. So a dilute solution will have a high water potential, a concentrated solution (with lots of dissolved solute) a lower water potential.
Osmosis is the
- net movement of water
- through a partially permeable membrane
- from a solution with a high water potential (a dilute solution) to a solution with a lower water potential (a concentrated solution)
Biological examples
Diffusion
- Oxygen diffuses from the air in the alveolus into the blood
- Carbon Dioxide diffuses from the air spaces in the leaf into the palisade mesophyll cells of the leaf
- Glucose diffuses from the blood into an actively-respiring muscle
Active Transport
- Nitrates are pumped from the soil into root hair cells by active transport
- In the kidney, glucose and other useful molecules are pumped from the nephron back into the blood by active transport.
- In nerve cells, sodium and potassium ions are pumped across the cell membrane to set up the gradients needed for a nerve impulse
Osmosis
- Water enters root hair cells from the soil by osmosis
- In the kidney, water is reabsorbed from the nephron by osmosis.
- In the large intestine, water is reabsorbed from the colon back into the blood by osmosis
There are many many more examples of each process, but this should be enough to be going on with…….