Category: Section 4: Ecology and the Environment
Carbon Cycle – Grade 9 Understanding for IGCSE Biology 4.10
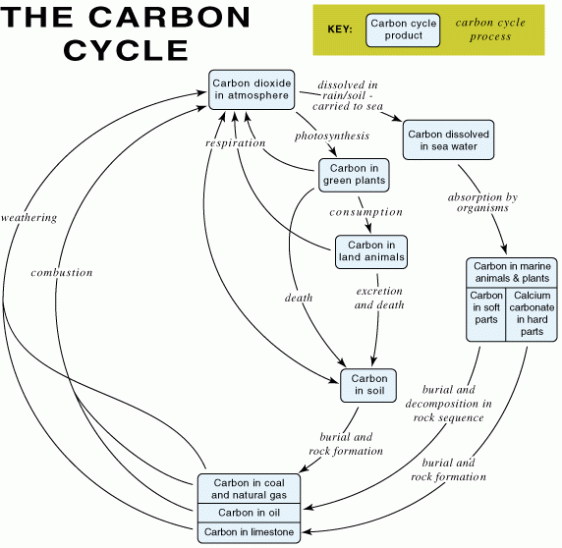
The Carbon cycle should really be much simpler to understand than the Nitrogen cycle I posted about yesterday. This is because the processes involved in moving carbon atoms from one compartment to the next in an ecosystem are more straightforward. There are four processes mentioned in the specification and you need to make sure you understand each.
- Photosynthesis: only happens in producers, takes CO2 from the air to produce complex molecules (carbohydrates/proteins/fats) that can be passed up food chain.
- Respiration: happens in all organisms (producers, consumers, decomposers) and turns carbohydrates into carbon dioxide
- Combustion: fossil fuels and plants can be burnt for fuel releasing carbon dioxide into the atmosphere
- Decomposition: two types of decomposition – in aerobic conditions decomposer organisms (bacteria/fungi) convert complex molecules in faeces/dead organisms into carbon dioxide: in anaerobic conditions, dead organisms can be turned into fossil fuels.
If you want to draw a carbon cycle from scratch to check you understand it, follow the procedure below.
1) Draw the following boxes showing where carbon atoms are found in an ecosystem – CO2 in air, carbon compounds in plants, carbon compounds in animals, fossil fuels and limestone, detritus in soil
2) Draw arrows linking the boxes with the following labels: photosynthesis, respiration, feeding, combustion, death and decay, death and no decay
That’s about as complicated as it gets.
Warning: Do not under any circumstances draw an arrow from the detritus in the soil directly to plants. Plants do not absorb any carbon containing molecules from the soil into their roots. Honestly, please believe me they don’t however much you want them to…. It would make the cycle more straightforward but they don’t – sorry. The only carbon-containing molecule plants absorb from their environment is CO2 and that as you all know is absorbed from the air in leaves in the process of photosynthesis.
Zondle quiz on carbon cycle to follow in due course: keep working hard! (I am jealous of those of you in Portugal at the moment although weather has been fine today in Northants!)
Bacteria of the Nitrogen Cycle: Grade 9 Understanding for GCSE Biology 4.11B
Here is my paragraph about each of the four sets of bacteria involved in the Nitrogen Cycle. 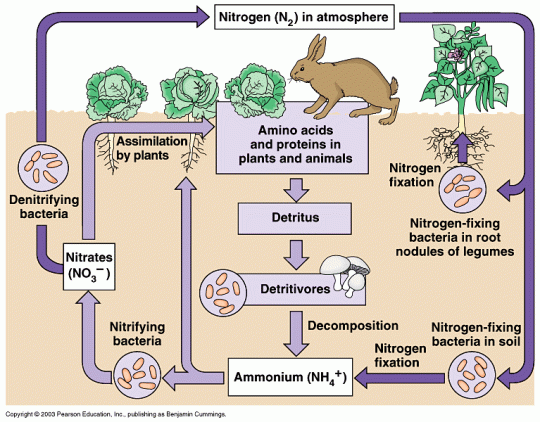
1) Decomposers (Putrefying Bacteria) These aerobic bacteria live in the soil. When an organism dies decomposers digest the proteins and DNA that are found in the cells of the organism and produce ammonium ions (NH4+) as a waste product. Decomposers will also break down the molecules in animal faeces and will decompose the urea in animal urine into ammonium ions.
2) Nitrifying Bacteria These are also aerobic bacteria that live in the soil. They get their energy by converting ammonium ions into nitrates (via an intermediate ion called a nitrite). Nitrifying bacteria are essential for the Nitrogen cycle because the nitrates they form are the ions that plants will absorb through their roots. Nitrates will be used by the plant to make amino acids, proteins and DNA and these can pass up food chains.
3) Nitrogen-fixing Bacteria The picture now gets a little more complicated. Nitrogen-fixing bacteria can convert atmospheric nitrogen found in air spaces in the soil into ammonium ions (see diagram above). These bacteria live in the soil and respire aerobically like all the bacteria mentioned so far. Some nitrogen-fixing bacteria have formed a symbiotic relationship with a family of plants that includes peas, beans and clover. These plants are called leguminous plants and have small nodules in their roots that contain the nitrogen-fixing bacteria. The relationship is a mutualistic one as both parties benefit. The plant benefits as these bacteria produce nitrate ions that the plant can absorb and use to make proteins and the bacteria benefit as they are protected from soil predators and have a stable environment in which to live.
 4) Denitrifying bacteria
4) Denitrifying bacteria
These are anaerobic bacteria that thrive in conditions where there is little oxygen in the soil. This often happens when the soil becomes water-logged so all the air spaces are flooded. Denitrifying bacteria are “bad news” for Nitrogen cycle as they get their energy by taking nitrates from the soil and converting them into nitrogen gas. This obviously reduces the nitrate available to the plants with roots in the soil and this is one of the reasons farmers like to keep soil well aerated for their crops.
I will post a quiz on Nitrogen cycle on Zondle later this evening and so if you want to test your understanding of this potentially tricky topic, I would suggest you have a go at my quiz. There are also plenty of past paper questions on Nitrogen Cycle in the red question booklet.
Nitrogen cycle for IGCSE Biology: Grade 9 Understanding 4.11B
I will make a blog post on each of the three “cycles” you need to know about for iGCSE. By far the most complicated is the Nitrogen cycle, so we might as well start there….
The first bit of understanding you need to is be clear the difference between how energy moves through an ecosystem and how matter (i.e. atoms) are exchanged between organisms and their environment. Energy in the ecosystems moves in a linear flow: there is no recycling of energy. The energy comes in at one end (in the producers through the process of photosynthesis) and is ultimately all lost as heat to the environment through the process of respiration. There is no possible way energy can be recycled. The “circle of life” that students like so much from Disney certainly does not apply here…. People find this idea very difficult to appreciate. All the time students will tell me that the energy in dead plants and animals goes into the soil and is then absorbed through the roots of plants: “it’s the circle of life sir” they earnestly tell me. And in the words of the late, great Amy Winehouse, I say “NO,NO,NO”…
Matter on the other hand is recycled through the ecosystem. The individual atoms that make up your body (H,O,C,N,S etc.etc,) have all been in other organisms and indeed will be again in the future. You took them in through your food and use these atoms to build the molecules that make up your cells. But ultimately all these atoms will leave your body either through metabolic processes or when you die and are decomposed. You could draw up a cycle for any of the atoms that are found in living things but your specification only requires you to understand two. How are carbon atoms cycled – the Carbon cycle – and how are nitrogen atoms cycled – the Nitrogen cycle. (You will also look at the Water cycle as well…..)
Things to understand about the Nitrogen Cycle:
1) Which molecules in living things contain nitrogen atoms?
Well the answer is fairly simple. Proteins are polymers of amino acids. Amino acids all contain an -NH2 group (amine group) and so Nitrogen is found in proteins. The bases in DNA (Adenine, Cytosine, Guanine and Thymine) are described as nitrogenous bases and so they contain nitrogen too.
2) Where are nitrogen atoms found in the ecosystem other than in the molecules of living organisms?
This is more complicated. Nitrogen gas makes up 78% of the atmosphere so clearly there is a lot of nitrogen in the air. In the soil, there will be urea from the urine of animals and urea contains nitrogen. There are also a range of ions found in the soil that contain nitrogen: the two most important are ammonium NH4+ and nitrate, NO3-. (I don’t know how to do subscript and superscript in WordPress and so you will have to excuse the rather ugly molecular formulae)
3) How do nitrogen atoms move from the abiotic (non-living) parts of the ecosystem and into the organisms?
There is only one way nitrogen atoms can move from the abiotic environment and into the organisms in an ecosystem. This is via plants that can absorb nitrate ions from the soil in their roots. This is a slight simplification but it will do at the moment. Look at the diagram above and find the arrow that shows assimilation of nitrates from the soil into plants.
4) How many different kinds of soil bacteria are involved in cycling nitrogen?
You need to know about four different kinds of bacterial that live in the soil that play a role in recycling nitrogen. Use the diagram above and your notes to describe the role each of these organisms play in the cycling of nitrogen atoms in the ecosystem.
- Decomposers (Putrefying Bacteria)
- Nitrifying Bacteria
- Nitrogen-Fixing Bacteria
- Denitrifying Bacteria
I am off to have my supper: another post about these bacteria will appear here later tonight or tomorrow. Please don’t read it until you have tried to write down a paragraph on each of the four types of bacteria in the bullet point list above.
Eutrophication – the least glamorous topic in IGCSE Biology 4.16 4.17
This is a topic it is easy to overlook in your revision: water pollution by sewage is hardly glamorous and when you combine it with the limited excitement of learning about fertilisers, you don’t have to be a genius to see why many students leave it out. But examiners seem to like eutrophication so it is worth making sure you are going to score full marks on any question they set.
Starting point in your understanding for this topic should be that what limits plant growth in many circumstances, and often in aquatic ecosystems, is the availability to the plant of nitrate ions. A nitrate ion (NO3-) is essential for the plant to make amino acids, and hence proteins and also DNA. Cells need more DNA and proteins to divide and grow so farmers spray extra nitrate ions onto their fields in fertilisers.
Nitrate ions are very soluble in water so if it rains, they can easily dissolve as the rain water passes through the soil a process known as “leaching”. These nitrate ions can thus end up in freshwater, for example streams and ponds where they have exactly the same effect as in the soil – they cause excessive plant growth. The plants in freshwater are often types of algae and if you have far too much nitrate in the water, this can cause an algal bloom. This excessive growth of plants is called eutrophication (or more properly hypertrophication)
Now the eutrophication story then proceeds like this….
If there is an algal bloom, this can eventually cover the surface of a pond so that light doesn’t penetrate to the multicellular plants that live on the bottom, the beautifully named “bottom-dwellers”. No light for these plants means they can’t photosynthesise and so they die. In the water there will be aerobic bacteria that act as decomposers and so if there is loads of dead material in the water, the populations of these bacterial decomposers will rapidly rise to break down the detritus. These bacteria remove dissolved oxygen efficiently from the water as their numbers go up (remember they are aerobic bacteria), so water quality rapidly falls. The lowering oxygen concentration in the water will itself cause animals from small invertebrates to larger fish to die and so a vicious circle is set up: less oxygen = more dead organisms = more decomposers = less oxygen.
The consequences for a pond in these conditions are very damaging. The complex ecosystem involving producers, consumers, food chains and so on is replaced by one with algae, decomposing bacteria and a few organisms that can survive in anaerobic conditions.
Anything that increases the numbers of decomposers in a freshwater ecosystem can cause eutrophication and the consequences described. The commonest cause is agricultural run off from fertilisers (nitrates/phosphates etc.) but untreated sewage can cause similar consequences. The decomposers need to break down the sewage, releasing nitrate ions into the water that cause eutrophication. The murkiness of the water can also kill bottom-dwellers directly and set the whole cycle off.
Eutrophication is a sequential process and often examiners use this question to test your ability to organise your answer properly. I would always answer a four or five marker on this topic with bullet points rather than a paragraph of text. Why do you think this is?
Now make a summary diagram to show eutrophication and answer a few IGCSE questions from the booklet.
Work hard!