Air Pollution part 2: Grade 9 Understanding for IGCSE Biology 4.12
It’s a cold February day in half term as I write this and I have already lit my log-burner in the living room downstairs. When this was fitted a year or two ago, I was told it is now a legal requirement in the UK to also fit a detector for one atmospheric pollutant that can be produced as a waste product of combustion of biofuels. This post is about that pollutant, carbon monoxide.
Carbon Monoxide
Carbon monoxide is a colourless, odourless gas. It is produced whenever carbon-based fuels (coal, gas, wood, charcoal) burn in an atmosphere with a restricted oxygen supply. The structure of a molecule of carbon monoxide is very simple: it is a carbon atom covalently bonded to a single oxygen atom.
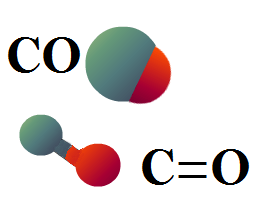
(My log burner has a control where you restrict air getting into the stove so that your massively expensive logs burn a little more slowly – hence the need for the CO detector) Carbon Monoxide poisoning results in 200 hospital admissions per year in the UK and around 40 deaths.
Why is carbon monoxide such a deadly gas?
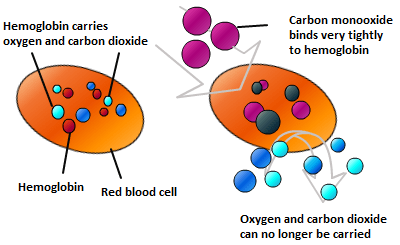
The symptoms of carbon monoxide poisoning are similar to flu. Tiredness, vomiting, headache, stomach pain…. They are caused because when carbon monoxide is breathed in, it will diffuse into the blood in the alveoli of the lungs. Carbon monoxide binds to haemoglobin, the protein found in red blood cells that transports oxygen. It binds about 70 times as effectively to haemoglobin, forming a stable compound called carboxyhaemoglobin. So it prevents haemoglobin molecules from transporting oxygen (and indeed carbon dioxide) around the body.
I hope that post is useful when you come to revise this section. It also links in of course to the topic on blood and indeed the health consequences of cigarette smoking. I’m sure I have posts on these topics as well to read if you are interested….. I’m off now to get my chimney swept and to check the batteries in my Carbon Monoxide detector.